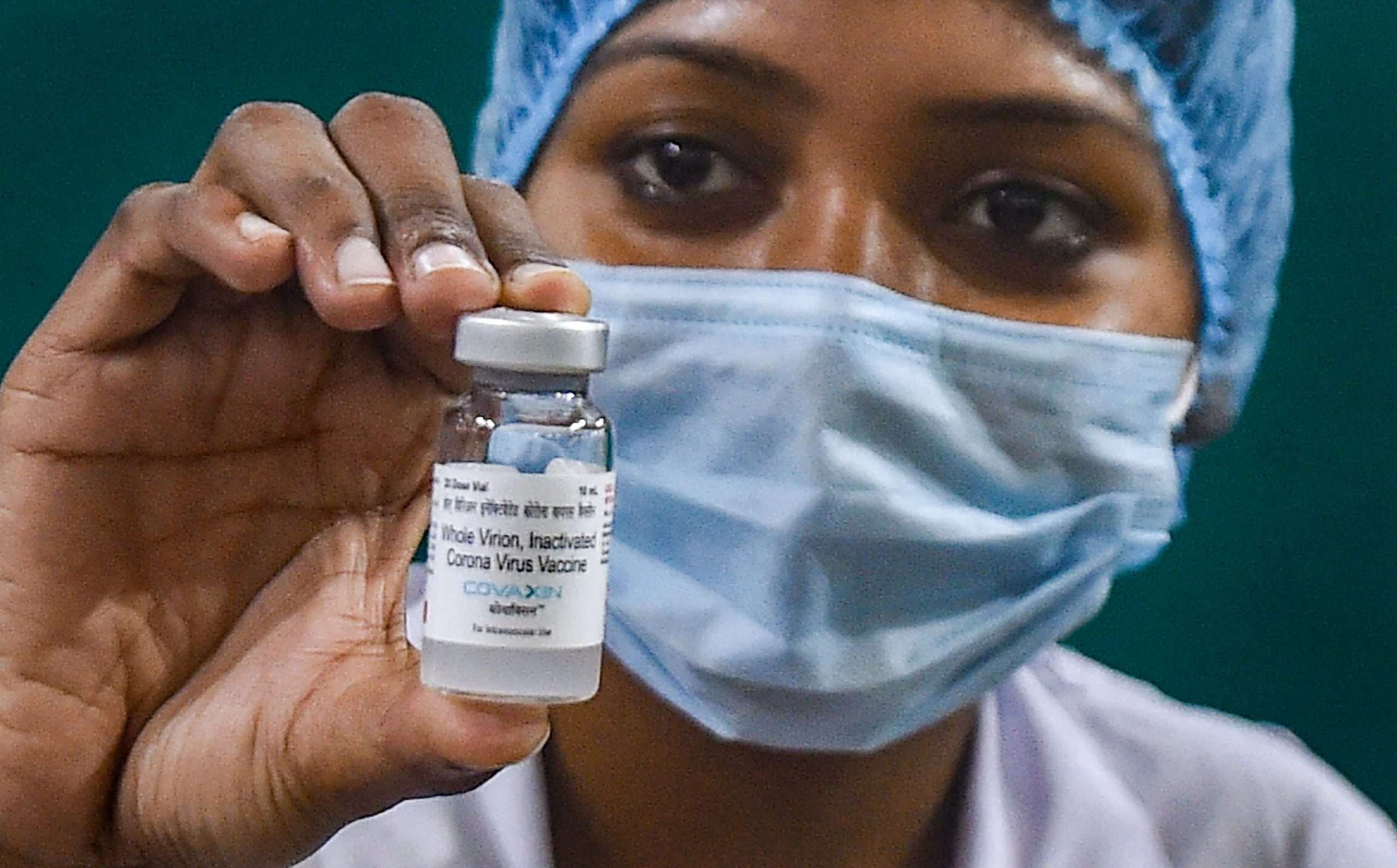
India’s only indigenously developed COVID-19 vaccine developed Bharat Biotech, Covaxin, is likely to approved by World Health Organisation (WHO) for an emergency use listing in the next four to six weeks, said chief scientist Dr Soumya Swaminathan.
Addressing a webinar, Swaminathan said a process is to be followed for approval and companies submit their safety data, complete trial data, and even manufacturing quality data for approval. She added that Hyderabad-based pharma giant has already started submitting the data and the dossier is being assessed. It is the next vaccine to be reviewed by our committee.
Also Read | What causes blood clots after COVID vaccination? Expert answers
She had recently said that the phase 3 trial data of Covaxin “looks good” and meets the safety profile of the international public health agency so far.
According to the phase-3 data, Covaxin has been found to be 77.8% effective against symptomatic COVID-19.
Also Read: Brazil-Bharat Biotech’s COVID vaccine deal that ran into trouble: Explained
16 countries, including Brazil, Mexico, Philippines and Iran, have already given emergency use authorisation to Covaxin. The WHO approval, if granted, will come as a relief to Indians and citizens of at least 12 countries where the vaccine is being administered.
The chief scientist also discussed the recent data on anti-parasitic drug Ivermectin as a possible COVID-19 treatment. The quality of studies were poor so when we put it in meta-analysis they did not suggest any evidence to use Ivermectin. The drug should be used only in clinical trials. There are a lot of medicines being used without evidence, she said.
Also Read| Will India be able to vaccinate its adult population in 2021?
India has been administering Covaxin since the beginning of its vaccination drive, which began on January 16, 2021. The Centre is confident and promising to vaccinate its entire adult population by the end of 2021.